Independent Clinical Laboratory Coding: CPT® Codes & Billing
Independent clinical laboratories CPT coding & billing: Current Procedural Terminology codes, payer reimbursement, administrative service codes, medical services.


Cameron Fletcher
Head of Growth at PayerPrice
Your lab client's revenue is shrinking and the explanation from their billing team is not holding up. CO-252 denials are climbing, the molecular panel they invested in last year keeps coming back denied even though the science is sound, and the hospital outreach line of business is collecting less per accession than the contract says it should.
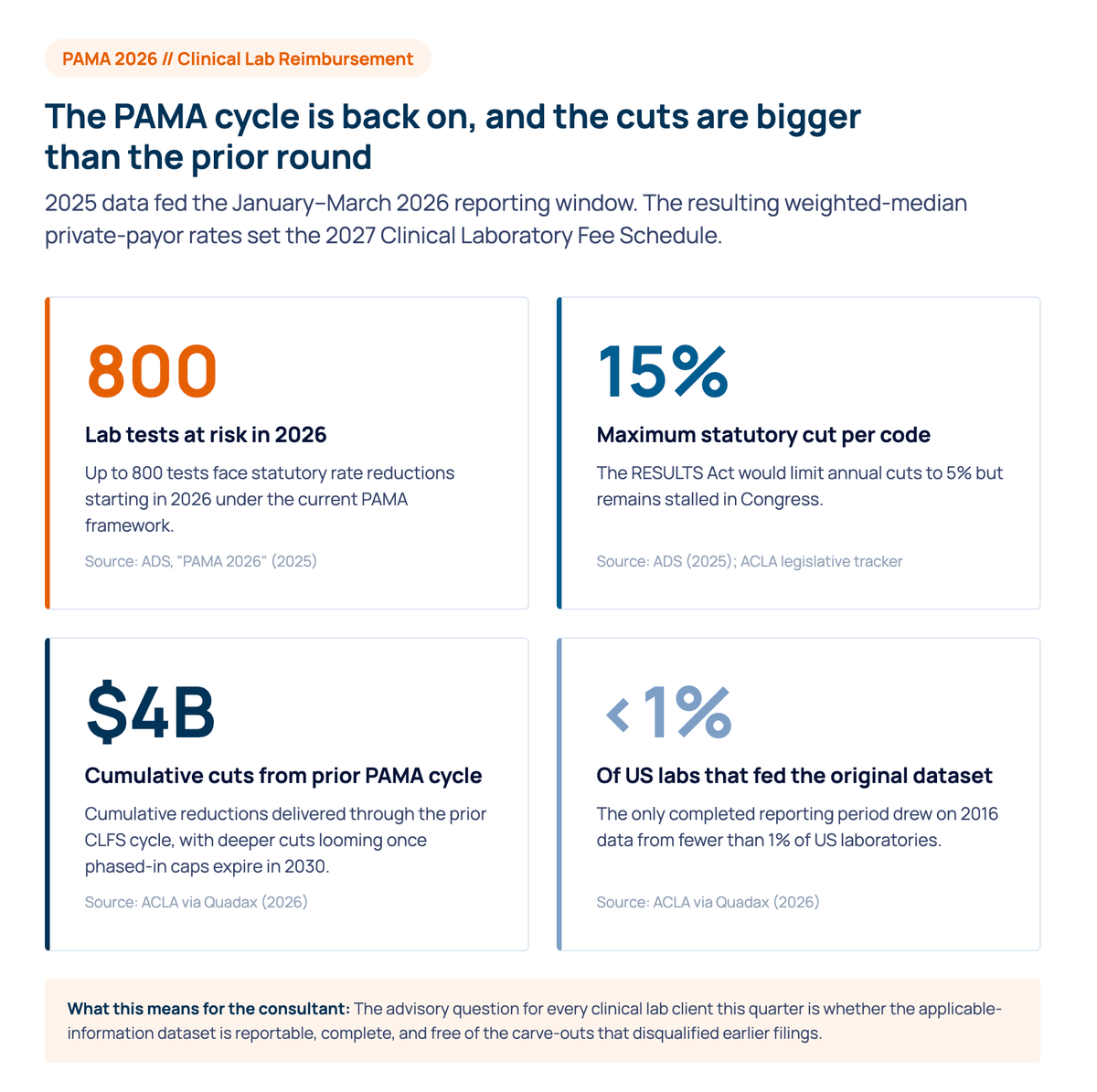
According to ADS, up to 800 lab tests face up to 15 percent reimbursement reduction starting in 2026, and the RESULTS Act remains stalled in Congress. According to Tom Scheanwald of APS Medical Billing, speaking to CAP TODAY, denial volume has risen and turnaround time on full payment has lengthened significantly. According to Robert L. Michel of Dark Daily, the FDA Final Rule on Laboratory Developed Tests, finalized CLIA revisions, and expanded MolDX Z-code policies are now in play simultaneously.
This clinical laboratories coding guide is built for the consultant who has to translate that regulatory tape into advisory action for every healthcare provider on their roster. It covers the 80000-series CPT code structure, modifier discipline, panel arithmetic, and the administrative service codes every lab biller knows, plus the four 2026 issues the existing first-page results skip: the PAMA private payor reporting cycle, the molecular pathology coding hierarchy and the MolDX Technical Assessment workgroup, and the 14-day rule for hospital outreach laboratory service billing.
What changed for independent clinical laboratories in 2026
The 2025 PAMA private payor reporting cycle, the FDA's final rule on laboratory developed tests, finalized CLIA revisions, and expanded MolDX Z-code coverage have all landed on independent clinical laboratories in the same year, making 2026 the first year laboratory coding decisions are inseparable from regulatory strategy.
The four shifts, in the order they hit the P&L:
- PAMA private payor reporting reopened. Q1 to Q3 2025 was the data collection period; January 1 to March 31 2026 is the reporting window; the resulting weighted-median rates set the 2027 Clinical Laboratory Fee Schedule. According to ADS, up to 800 tests face up to a 15 percent statutory cut. The American Clinical Laboratory Association has documented that the only completed reporting period drew on 2016 data from fewer than 1 percent of U.S. laboratories. Confirm every client's applicable-information dataset is reportable and free of disqualifying carve-outs.
- The FDA Final Rule on Laboratory Developed Tests. Phase-in began May 2024 and runs through 2028 in five stages. Stage 4 brings premarket review for high-risk LDTs in 2027. Consultants need a position on which assays remain in scope, which qualify for grandfathering, and how the rule interacts with FDA approval pathways and the lab's PLA and MolDX strategy.
- CLIA revisions. The Centers for Medicare and Medicaid Services finalized updates to personnel qualifications and proficiency testing in late 2024. POL clients now sit inside an active national audit posture, and eligibility for CLIA-waived test use depends on staying current with the requirements.
- MolDX expansion. Palmetto GBA, Noridian, CGS, and WPS continue to expand LCD scope, including respiratory multi-pathogen panels and tuberculosis subtype detection workflows. Robert L. Michel called it the "perfect storm" framing in 2024: flat federal spend, rising compliance load, and narrowing margins on services and procedures across the lab P&L. Each commercial payer reimburses the same code differently, and how each health plan responds to these shifts is the variable a consultant has to model.
Clinical laboratory CPT codes, modifiers, and administrative service codes
Clinical lab coding rests on four moving parts: the CPT 80000-series structure, modifier discipline, panel arithmetic, and ICD-10 diagnosis codes alongside administrative service codes. Consultants who QA only the procedure code without QA-ing the modifier rules will miss most of the denial drivers.
The CPT 80000-series breaks into seven families: organ-and-disease-oriented panels (80047 through 80076), drug assays, evocative testing, chemistry, hematology and coagulation, immunology, microbiology, and cytopathology. Within microbiology, infectious disease NAAT codes (87471 through 87653) carry the highest reimbursement and the heaviest payer scrutiny. PCR and amplified probe technique tests for tuberculosis, RNA-based viral detection, and other pathogen subtype identification fall here. Each laboratory test has its own analyte definition that determines whether the test performed is reportable as one unit or as a multi-component panel.
The HCPCS Level II codeset complements CPT for laboratory claims. G-codes (drug testing) and J-codes (biologicals) sit in HCPCS rather than CPT. Always confirm whether a CPT or HCPCS code is the correct one for the test performed and that codes are submitted on the right claim form.
The lab modifier set most often misapplied:
- 91 marks a repeat clinical diagnostic test on the same date for clinical reasons, not re-runs of failed tests.
- 90 marks the outside or reference lab on a referring lab's claim, paired with the CMS 30 percent rule.
- 59 versus the X{EPSU} subset (XE, XS, XP, XU). Audit which split each MAC and commercial payer expects.
- QW identifies a CLIA-waived test (for example 87880 versus 87880-QW).
- GA, GZ, GY govern Advance Beneficiary Notice posture for non-covered or self-pay tests, including patient deductible scenarios.
Panel arithmetic is the most undertaught topic in lab coding. CMS Chapter 16 specifies that automated profile and organ-disease panels pay the lower of billed charges, the panel fee, or the sum of components. The most common unbundling trap is reporting BMP (80048) plus a non-included analyte separately when CMP (80053) was the correct single code, triggering an NCCI procedure-to-procedure edit. Add the National Correct Coding Initiative Medically Unlikely Edit table (the maximum units of service per code per day) to the LIS edit layer rather than the appeal layer. NCCI edits and the unlikely edits subset are the front line against incorrect coding and bad code combinations.
ICD-10 specificity is the universal medical-necessity denial driver. Diagnosis codes must support the medical services rendered, and interpretive review at the front end catches the high-dollar specialty codes before submission. Specimen collection codes (36415 venipuncture and 99000 handling) carry their own travel-allowance economics for hospital outreach.
No results
Adjust your filters or try a different code.
The CPT® codes editorial panel, PLA workgroup, and HCPCS coding system
The CPT® codes editorial panel of the American Medical Association (AMA), the Pathology and Laboratory Coding Caucus, and the PLA Technical Advisory Group are the three workgroup bodies that decide which laboratory tests get a Current Procedural Terminology code. Consultants who track their meeting calendars are the first to see code assignments before they hit the lab.
The CPT Editorial Panel approves Category I, II, and III codes through quarterly meetings. The Pathology Coding Caucus drafts new pathology and laboratory codes for editorial panel review. The PLA Technical Advisory Group governs the Proprietary Laboratory Analyses (PLA) codes, the alphanumeric "U" codes single-source labs use for proprietary assays. The HCPCS coding system, maintained by a Centers for Medicare and Medicaid Services workgroup, sits alongside CPT.
Three things make this layer matter for a medical practice operating an independent clinical laboratory:
- Quarterly PLA releases. Codes are submitted by labs and IVD manufacturers through the AMA's CPT Smart App. The PLA workgroup reviews clinical utility, peer-reviewed validation, and FDA approval status.
- Editorial panel migration of high-volume PLA tests to Category I. When a PLA test reaches sufficient volume and evidence, the editorial panel can promote it. Reimbursement implications are immediate.
- Health plan medical policy alignment. Payers and health plans set medical policy off the AMA codeset and HCPCS. A new code with no medical policy entry behaves like an unlisted code in adjudication. A qualified health care consultant should track the gap between code release and medical policy update at every commercial payer.

The molecular pathology coding hierarchy and MolDX Z-codes
The molecular pathology codeset is hierarchical. Report the most specific code available in this order: PLA, then Tier 1, then Tier 2 or genomic sequencing procedure, then MAAA, then 81479 unlisted. Overlay the MolDX Z-code requirement for any test billed in a Palmetto, Noridian, CGS, or WPS jurisdiction.
The five-rung hierarchy:
- PLA codes (0001U through 0500U-plus). Sole-source or multi-provider proprietary tests. The AMA releases new codes quarterly. PLA always takes precedence over an unlisted code. As Sobihaa Selvaraj of SYNERGEN Health wrote for AAPC in August 2025: "Whenever there is a PLA code for a specific proprietorship-based test panel, billing that code takes precedence over unlisted molecular pathology codes."
- Tier 1 (81105 through 81383). Single-analyte molecular procedures with assigned CPT codes.
- Tier 2 (81400 through 81408). Analyte tiers based on technical complexity. Tier 2 cannot be self-assigned.
- Genomic Sequencing Procedures (next-generation sequencing or NGS panels) and Multianalyte Assays with Algorithmic Analyses (81410 through 81471, 0002M through 81599). Multi-gene panels with strict gene-list semantics and algorithmic MAAA codes split between Category I MAAA and Administrative MAAA in CPT Appendix O.
- Unlisted molecular (81479) and unlisted chemistry (84999), microbiology (87999), and cytopathology (88299).
Common molecular workflows: amplified probe technique PCR for infectious disease and tuberculosis, RNA viral load quantification, oncology somatic-mutation NGS panels, and pathogen subtype identification. Each maps to a different rung, and panel testing menu decisions follow the rung the assay lands on.
The MolDX Z-code requirement is the second axis. For molecular tests billed in Palmetto, Noridian, CGS, or WPS Medicare Administrative Contractors jurisdictions, the lab must obtain a DEX Z-code from the Palmetto MolDX program and submit it on the claim alongside the CPT. Without an active Z-code, the test is treated as not medically necessary regardless of clinical utility. Lighthouse Lab Services described the operational reality:
"If a lab has not yet received a CPT Code from MolDX along with a defined reimbursement, the lab should anticipate seeing those services denied by Medicare. Because the panel being performed is not already FDA approved, MolDX requires that the clinical utility and validity of the panel be proven through a Technical Assessment process. Until that time, the panel is considered to not be medically necessary, and appeals are not seen to be helpful."
- Lighthouse Lab Services RCM team
The Technical Assessment process means clinical-validity dossier preparation, peer-reviewed evidence review, and coverage determination. MolDX additional requirements often include analytical validation and detectable lower-limit-of-detection data for the assay. New PLA codes also enter without a payer price, and commercial payers trail the MAC gapfill or crosswalk by six to twelve months. ADLT designation earns 9 months of actual-list-charge pricing followed by weighted-median private-payor pricing.
The fifth axis is the payer-specific genetic-testing prior authorization program. eviCore by Evernorth covers many commercial plans. Avalon Healthcare Solutions covers most Blue Cross Blue Shield plans. Concert Genetics, now operating with Cohere, covers Humana. Optum covers UnitedHealthcare. Each program has its own laboratory benefit management criteria and registration workflow.
Ready to see how your rates compare?
PayerPrice gives you instant access to negotiated rate data across payers and providers so you can benchmark, negotiate, and optimize with confidence.
Hospital outreach laboratory service billing under the 14-day rule
The 14-day rule, also called the Date-of-Service exception under 42 CFR 414.510(b)(5), shifts billing responsibility for molecular pathology and ADLTs ordered within 14 days of a hospital outpatient discharge from the hospital to the performing lab. Most hospital outreach billing operations get it wrong, and most consultants find recoverable dollars on the first audit pass.
The 2018 amendment carved molecular and ADLT laboratory tests out of OPPS packaging when ordered after the patient leaves the hospital, and required the performing lab to bill Medicare directly under the Clinical Laboratory Fee Schedule. The rule does not apply when the patient remains inpatient: inpatient laboratory tests flow through the DRG and are not separately reportable. Valerie Rinkle, MPA, of HCPro:
"The 14-day rule, when it comes to outpatient molecular pathology and ADLTs, is no longer applicable for outpatients. You completely separate that from the outpatient encounter and the performing lab must bill, because if you look at those criteria, there's pretty much no outpatient account that isn't going to meet those criteria."
- Valerie Rinkle, MPA, HCPro
The implications:
- Bill type 14X for hospital outpatient hospital reference work.
- Place of Service 81 (independent laboratory) on the performing lab's CLFS claim when the outpatient ordering condition is met; POS 22 for hospital outpatient.
- The hospital should not include the molecular or ADLT charge on its OPPS claim. When laboratory claims are submitted on the wrong bill type, the recovery path is appeal, not rework.
- Patient eligibility checks at order entry should flag the outpatient discharge date so codes are submitted on the right claim by the right entity.
The 90-day audit path: pull molecular and ADLT send-outs from the LIS, confirm date of service against the patient's discharge date, confirm whether the performing lab billed CLFS, and reconcile against the contracted rate. Recoverable dollars cluster in the 5 to 10 percent of accessions on the boundary of the 14-day window.
Closing: a seven-line audit for your next clinical laboratories engagement

Run this on the next remit and PAMA reporting plan, in this order:
- Confirm the lab's PAMA applicable-information dataset is reportable and free of disqualifying carve-outs.
- Map the assay menu against the FDA LDT rule phase-in and identify Stage 4 high-risk items.
- Pull the QW inventory and reconcile against the Certificate of Waiver scope.
- Sample 30 days of MolDX-jurisdiction molecular claims and confirm active Z-code coverage.
- Audit a 90-day sample of molecular send-outs against the 14-day rule.
- Pull the top 10 high-dollar codes and reconcile contracted rate versus paid rate per payer and per health plan.
- Track first-pass clean claim rate and denial rate by reason monthly.
The fundamentals in any clinical laboratories coding guide are not wrong. They are insufficient for the consultant who has to translate them into advisory action with the lab CFO. The codeset is the surface; the regulatory tape and the contract economics are what the consultant is actually being paid to fix.
PayerPrice is the contract analytics and underpayment-recovery layer that sits behind the coding function, with a rate benchmarking platform that surfaces underpayments off the 835 remittance. If the audit surfaces leaks you want to instrument and recover at scale across the lab client's payer mix, see how PayerPrice supports contracting consultants.
Share this article
Help spread the knowledge by sharing with others
Ready to see how your rates compare?
PayerPrice gives you instant access to negotiated rate data across payers and providers so you can benchmark, negotiate, and optimize with confidence.
Related Articles
Continue exploring healthcare transparency and compliance topics
Multi-Specialty Coding Guide: Billing in Multi-Specialty Practices
A multi-specialty coding and billing guide for physician groups. Covers CPT, modifier 25, NPP rules, and fee schedules in multi-specialty practices.
Ophthalmology & Retina Coding Guide: Billing & Coding
2026 ophthalmology and retina coding guide for the retina practice. CPT codes, billing and coding, diagnostic imaging, cataract, ophthalmic reimbursement.
2026 Oncology Coding Guide for Physician Groups
The 2026 oncology coding guide for community practice owners. ICD-10 sequencing, chemo CPT hierarchy, drug J-codes, and where revenue actually leaks.